This is a OPINION PIECE and should be interpreted with CAUTION!
The text is ‘overtly simplified’ for general audience and DOES NOT CLAIM scientific accuracy but is only intended to present easy to understand information.
first published : 26.05.2021
What we know about the Delta SARS-CoV-2 variant (mutated in India) and why it is a concern?
The new SARS-CoV-2 lineages from India which are the likely cause of exceptional rise in new cases (and deaths) were first detected in Dec 2020 and currently categoried into three main sub-variants, the B.1.617.1, B.1.617.2 and B.1.617.3, each with distinct mutations in addition to shared mutations. The B.1.617.2 is a ‘variant of concern‘ and recently recommended a greek alphabet nomenclature (Delta SARS-CoV-2 varaint for B.1.617.2) according to WHO. By early May 2021 these varaints lead to an explosive number of of over 400,000 new cases and 4,000 deaths each day for several days, resulting in hospital service capacity exhaustion and oxygen supply shortages.
Following is a summary of the mutations and there effects (no particular order) for with data sources. For other mutations, still articles are searched:
- L452R > increased transmissibility and vaccine induced immunity evasion (medrxiv, 9th March 2021, article in pre-print, PDF file, 3.3Mb)
- E484Q > vaccine induced immunity evasion (The LANCET Microbe, 7th Apr 2021, PDF file 435kb)
- D614G > increased transmissibility (Nature, 26 Oct 2020, PDF file 5.2Mb)
These varaints, as expectedly, transmitted to many parts of the world. Notably, in UK, the sudden rise in cases are mostly associated with B.1.617.2 variant (although all three sub-varaints has detected so far), reasoned mostly due to travel to/from India and the following community transmission. Currently UK has designated the B.1.617.2 lineage as a ‘variant of concern’ (as of 19th May 2021).
Are these SARS-CoV-2 variants more transmissible?
An advisory committee to the UK government named ‘Scientific Pandemic Influenza Group on Modelling’ (SPI-M-O), reported on 12th May 2021, “SPI-M-O is confident that B.1.617.2 is more transmissible than B.1.1.7, and it is a realistic possibility that this new variant of concern could be 50% more transmissible” (text from the report, PDF file, 612kb). In later text (same report), “the principal result, that B.1.617.2 is growing more quickly than B.1.1.7, is robust to these sensitivities“.
Similarly, Prof. Wenseleers, Department of Biology, Katholieke Universiteit Leuven, Belgium, reported on 18th May 2021, that in the UK, the Delta variant is rapidly spreading and that it has a growth rate advantage of 13% per day over the Alpha varaint (B.1.1.7).
Also, as mentioned above with regard to the mutations, all three variants has the L452R and D614G mutations, both associated with increased transmissibility.
Together, it is evident that the Delta varaint (B.1.617.2) if not the other two as well, is very likely to be ‘more transmissible’ than the Alpha variant (B.1.1.7).
Can these SARS-CoV-2 variants be detected by current PCR and rapid antigen test kits?
There is very limited data on this topic for clarification, but since there are no major deletion or insertion of genomic sequences, all three varaints are expected to be detected by the standard diagonistic PCRs available.
As for the rapid antigen tests kits, it is very difficult to make any comment. There are hundreds of different rapid antigen test kits available and without proper screening, it cannot be determined if they can detect the these three varaints or not.
Can these SARS-CoV-2 variants escape current vaccine induced immunity?
Although the SARS-CoV-2 varaint B.1.617.2 is spreading fast in some parts of the UK and probably ‘other unconfimrmed’ parts of Europe as well, the numbers are still low (in terms of sequencing confirmed cases), and in the SPI-M-O report, the committee noted that “There are local areas in all nations where the epidemic is increasing and some localities, such as the North West and Bedford, have fast growth in cases of S-gene positive variants, including B.1.617.2“, which in light of the over 72% of the UK adult population has already received at least the first dose of a COVID-19 vaccine, data as of 26 May 2021, it may be perceived that the these varaints partly escape current vaccines. Of course more data and details are need before further confirmation, but the first indications point towards these ‘the three varaints that originated in India, especially the B.1.617.2 can escape from the current COVID-19 vaccine induced immunity’.
The same report also cautions “There is an inherent lag between vaccination and the establishment of protection of the vaccinated individual, and B.1.617.2 has the potential to spread very rapidly out of areas where it is currently present.”
It might also be of interest to mention that the sub-varaint, B.1.617.1, has an additional mutation called 484Q, which (the mutant site) has been reported earlier for strong association with vaccine escape (Nature, Published 11 March 2021, title: Sensitivity of SARS-CoV-2 B.1.1.7 to mRNA vaccine-elicited antibodies, PDF, 12.6Mb).
In an another research artcile, currently in pre-print (not peer reviewed yet), authors from the Public Health England, UK, reported only 33.5% vaccine effectiveness against B.1.617.2 varaint after first dose, 87.9% vaccine effectiveness after two doses of BioNTech-Pfizer COVID-19 vaccine and 59.8% vaccine effectiveness after two doses of AstraZeneca COVID-19 vaccine (PDF file, 304kb).
This press release from CureVac on 16.06.2021 is very important as it gives the first direct evidence that the mRNA based vaccines against the original SARS-CoV-2 genome, and hence called the first-generation mRNA vaccines, are less effective against the currently circulating evolved SARS-CoV-2 genomes (also known as the alpha, beta, gamma, and delta varaints, together known as the ‘variants of concern’). Principally all mRNA formulations work the same, that is the underlying ‘mRNA sequence’ translates into a single type of (spike) protein and consequently a single type of antibodies which may or may not be effective against a pool of varaints. This is exactly the situation in case of CureVac’s mearge 47% vaccine efficacy in Phase 2b/3 clinical trials, highlighted by 57% of the COVID-19 positive cases sequenced, being the ‘varaints of concern’, which are likely escaped the mRNA vaccine induced immunity!
Are these SARS-CoV-2 variants already in Europe / Luxembourg?
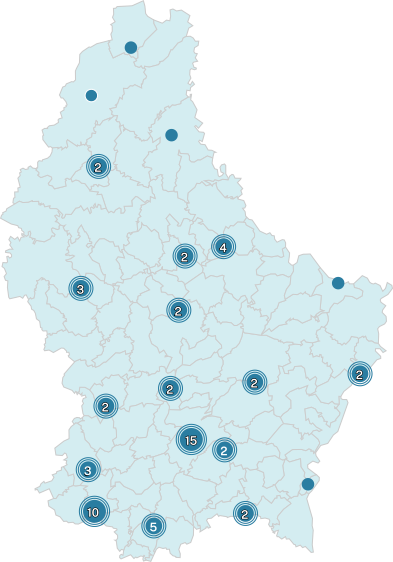
According to the cov-lineages.org which uses SARS-CoV-2 genome sequence data submitted to GISAID, a global repository of open-access genomic data, at least 53 countries globally have confirmed prescence by sequencing of these varaint(s) as of 26.05.2021, including Luxembourg (5 and 1 sequences of B.1.617.1 and B.1.617.2, respectively).
first published : 05.06.2021
Summary of SARS-CoV-2 ‘variants of concern’ and their first sampling in Luxembourg.
| PANGO lineage | WHO label | First detected – Country | First detected | functional mutations | Transmissibilty* | Effect on Immunity | In Luxembourg** |
|---|---|---|---|---|---|---|---|
| B.1.1.7 | Alpha | UK | Sep. 2020 | N501Y, D614G, P681H | Yes | No | Yes, Dec 2020 |
| B.1.351 | Beta | South Africa | Sep. 2020 | K417N, E484K, N501Y, D614G, A701V | Yes | Escape | Yes,, Jan 2021 |
| P.1 | Gamma | Brazil | Dec. 2020 | K417T, E484K, N501Y, D614G, H655Y | Yes | Escape | Yes, Mar 2021 |
| B.1.617.2 | Delta | India | Dec. 2020 | L452R, T478K, D614G, P681R | Yes | Escape | Yes, Apr 2021 |
** data source: Laboratoire National de Sante, Luxembourg
First Insights on Pfizer-BioNTech COVID-19 Vaccine induced Immunity against the Varaints of Concern, Beta and Gamma.
The Pfizer-BioNTech COVID-19 vaccine is the most widely used vaccine across the EU (196 Million doses out of 297 Million doses distrubuted across EU as of 4th June 2021) and represents 68% of all vaccine doses available in Luxembourg (data as of 4th June 2021), making it a very important player in terms of its efficacy against the evolving SARS-CoV-2 varaints. Early indicator in this direction is now available in a research article (correspondence) in the Lancet, a world-renowned and leading medical journal, titled ‘Neutralising antibody activity against SARS-CoV-2 VOCs B.1.617.2 and B.1.351 by BNT162b2 vaccination‘, first published 3rd June 2021 (PDF file, 72kb).
The authors show that the vaccine induced immunity (SARS-CoV-2 spike protein neutralizing antibodies) is inversely related to age, but not with gender or body-mass index (Supple. Fig. 2, PDF file, 4Mb). In other words, with increasing age, the vaccine induced neutralizing antibodies are lower. Moreover, the decrease continued with time as well, meaning, the antibodies further decreased with time.
Together, this brings new challenges, most notably are:
- The Beta and Delta varaints are more transmissible than the Alpha and wild-type varaint
- The Beta and Delta varaints can escape vaccine induced antibodies
- The Pfizer-BioNTech vaccine induced antibodies decrease rapidly with age and time
This is a OPINION PIECE and should be interpreted with CAUTION!
The text is ‘overtly simplified’ for general audience and DOES NOT CLAIM scientific accuracy but is only intended to present easy to understand information.
The above study has two main possible outcomes. First, there would be a need for ‘updated’ vaccine in response to the evolving varaints, and meanwhile, older population might need a ‘third (consecutive) booster dose’ every 4-6 months.
Second, the vaccine efficacy on an individual (gender, preconditions, comorbidities, etc) or sub-population (age, work, etc.) would need continuous evaluation against the evolving varaints.